 Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. Our Rydberg equation calculator is a tool that helps you compute and understand the hydrogen emission spectrum.You can use our calculator for other chemical elements, provided they have only one electron (so-called hydrogen-like atom, e.g., He, Li², or Be³). In 1913, a Danish physicist, Niels Bohr (1885–1962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. 
Each line can be calculated from a combination of simple whole numbers. Chemistry spectrum, Line spectrum series, chemistry, chemistry Hydrogen emission spectrum, hydrogen absorption. The lines in the hydrogen emission spectrum form regular patterns and can be represented by a (relatively) simple equation. Where \(n_1\) and \(n_2\) are positive integers, \(n_2 > n_1\), and \( \Re \) the Rydberg constant, has a value of 1.09737 × 10 7 m −1. The origin of the hydrogen emission spectrum. Rutherford’s earlier model of the atom had also assumed that electrons moved in circular orbits around the nucleus and that the atom was held together by the electrostatic attraction between the positively charged nucleus and the negatively charged electron.\] It is the exact opposite of the emission spectrum Both emission and absorption techniques can be used to get the same information about the energy levels of an atom. Line spectra were first observed and studied in the latter half of the 19th century, but it was only in the 1920s that a full explanation was finally developed. By looking at the specific wavelengths of light that are either absorbed or emitted from a sample of H atoms, we discover something about the energy of the. Figure 12.9 shows the absorption spectrum. 3: When light from a hydrogen gas discharge tube is passed through a prism, the light is split into. The figure below shows the atomic emission spectrum of hydrogen. The spectrum of hydrogen is particularly important in astronomy because most of the universe is made of hydrogen. 
Rydberg's equation is as follows: 1 RH ( 1 n21 1 n22) (12.3.3) (12.3.3) 1 R H ( 1 n 1 2 1 n 2 2) where. The line spectrum of each element is so characteristic of that element that its spectrum may be used to identify it. Bohr’s model required only one assumption: The electron moves around the nucleus in circular orbits that can have only certain allowed radii. In Figure 12.8 you can see the line emission spectrum of hydrogen. An atomic emission spectrum is the pattern of lines formed when light passes through a prism to separate it into the different frequencies of light it contains. Three years later, Rydberg generalized this so that it was possible to determine the wavelengths of any of the lines in the hydrogen emission spectrum. 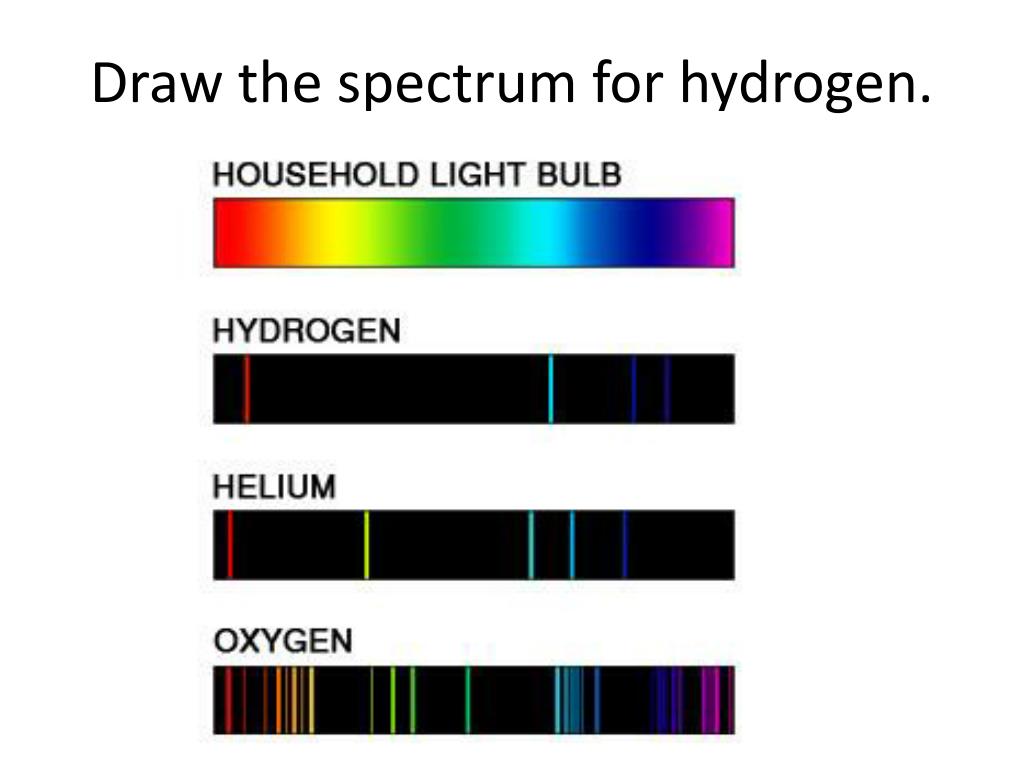 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
